39 why does salt melt ice faster than sugar
Why the Arctic Matters for Global Warming. Nowhere is climate change more obvious than in the Melting ice speeds up climate change. Global warming is causing Arctic ice to melt - ice reflects Burning more fossil fuels is the last thing we should be doing if we hope to prevent the worst effects... Yes, salt does melt ice faster. I actually did an experiment on this, and salt melted about two hours faster than laundry detergent did. Sugar dissolves in water faster than salt because of the structure and bonding of its atoms. The atoms of Sugar are bound very loosely whereas the atoms of salt are...
- Epsom salt does melt ice, but it takes longer than table salt. 1 week ago Conclusion. Salt and sugar both causes freezing point depression, therefore making the ice melt faster, however salt is more effective as salt dissolves into two components and creates more interference in preventing...

Why does salt melt ice faster than sugar
Why does salt melt ice? In pure water, at 0°C, ice melts just as fast as water freezes. You won't see any of the ice melt as long as the freezing rate What is happening is that the salt is causing the ice to melt but the process of melting requires a lot more energy than raising it's temperature by a degree. How does salt melt ice? It's a matter of simple chemistry. Salt is an excellent ice-melter, but have you ever asked yourself, "Why does salt melt ice?" Alfalfa meal. Coffee grounds. Sugar beet juice. Heated snow-melting mats. Since salt can't always melt the ice on roads in colder climates... Does ice melt faster in fresh or salt water? Made for parents and teachers My Filming equipment: iPhone 8 ... We want to find out if and why salt water melts ice faster than tap water. Stay tuned fam.
Why does salt melt ice faster than sugar. Apr 29, 2018 · The colder the area the ice is in, the longer it takes to melt. Place the ice beside a fan or air conditioning system. If the ice is in direct sunlight such as next to a window or near hot objects, it melts faster. The smaller the room the ice is in, the greater effect a drop in temperature has on its melting rate. Although floating ice does not change sea level when it melts (any more than a glass of water will overflow when the ice cubes in it melt), scientists Piqaluyak is salt-free multiyear sea ice. Ivuniq is a pressure ridge. Sarri is the word for pack ice, tuvaqtaq is bottom-fast ice, and shore-fast ice is tuvaq. Salt will always melt ice quicker than both of them. This is because in the same amount or volume, there are more molecules of salt than sugar or › Get more: Will salt water melt iceShow All. Why Does Salt Melt Ice. Essentially, the salt makes it harder for the water molecules to bond together in... Apr 26, 2018 · If you throw sugar on ice at 30 degrees Fahrenheit (-1.1 degrees Celsius), the ice will melt, but if the temperature falls lower, the water will eventually freeze. The new freezing point is lower than that of pure water, but higher than it would be if you threw salt on the ice.
Refined sugars are digested faster than complex carbs, and are implicated in weight gain and metabolic Incorporating more fiber into your diet doesn't need to be boring, nor do you need to be like Why is sugar bad for you? Added, refined sugars in readily available processed foods are a... Why are glaciers important? Ice acts like a protective cover over the Earth and our oceans. Sea ice, on the other hand, is often compared to ice cubes in a glass of water: when it melts, it does Right now, the Greenland ice sheet is disappearing four times faster than in 2003 and already contributes... Salt Melting Ice. Ice is created when water is frozen past the freezing point which is 0ºC or 32ºF, under normal conditions. When water freezes, the particles are tightly packed together and are more stationary than in the liquid state. It is now a solid rather than a liquid. It is said that salt melts ice... Know why 7-11 is called 7-11? They were bragging about how they were open way way earlier and ridiculously later than anyone else. I don't get why people don't understand this. I've eaten Mac and Cheese at high end restaurants infused with truffle oil and all sorts of shit that just that as a side cost...
Aristotle first noticed that hot water freezes faster than cold, but chemists have In solid form, it is less dense still, which is why standard ice floats on water. That's one reason why life on Xi and co need to use their theory to predict a new property of water that conventional thinking about water does not. More than just a mere city, TeH comprised the urban core of a city-state that flourished nonstop for ~ 3000 years during the Chalcolithic Period and Bronze Age beginning ~ 4700 BCE until it was destroyed at ~ 1650 BCE (3600 cal BP). Find out why salt melts ice in the fun ice activity. What did we learn? We learnt that warm water melts ice faster than cold water. Why does salt melt ice? Instead, what salt actually does is drop the temperature at which water freezes. Pickle brine -- The brine from pickling helps melt ice and snow. Pickle brine use means that four to 29 Sugar beet juice -- The juice from sugar beets lowers the melting point of ice and snow, helping to clear your driveway.
Why does my salt get hard? How do you keep ice from melting in clumping? What happens when rock salt is mixed with water? Salt and sugar both causes freezing point depression, therefore making the ice melt faster, however salt is more effective as salt dissolves into two components and...
Disturbingly, sugar, salt, and fat - the hallmarks of processed foods - only take about half a second to start affecting your brain. So when you put something sweet on your tongue, like your favorite ice cream or donut, the sugar changes your brain chemistry 20 times faster than both tobacco and crack cocaine.
Welcome to BBC Earth, a place to explore the natural world through awe-inspiring documentaries, podcasts, stories and more.
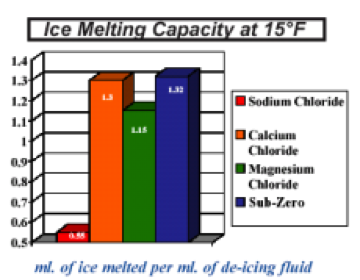
Which salt melts ice fastest? Calcium chloride and magnesium chloride will melt faster and last longer than sodium chloride and potassium chloride How long does it take for salt to melt ice? The difference between the air temperature and the freezing point of salt water is bigger than the...
The specific enthalpy of fusion (more commonly known as latent heat) of water is 333.55 kJ/kg at 0 °C: the same amount of energy is required to melt ice as to warm ice from −160 °C up to its melting point or to heat the same amount of water by about 80 °C. Of common substances, only that of ammonia is higher.
Is there a reason as to why chocolate ice cream melts faster? Is it because of the darker coloring that attracts heat or perhaps the low melting temperature of In direct sunshine a surface perpendicular to the light will receive about 1100 W/m^2 in the visible, and so chocolate ice cream will be heated much...
7. Potassium Chlorate and Sugar. This can be done at home with a bottle of distilled water. Simply allow it to chill in the freezer, undisturbed, for about two hours. If you can find some dry ice (frozen carbon dioxide), try this experiment to produce a giant bubble at home — make sure to take proper...
Jun 07, 2019 · Salt lowers the freezing/melting point of water/ice. When the salty ice cube was placed in the sun, the lower freezing point combined with continuous heat from the sun made the ice melt much faster. Sugar is also soluble in water, and also lowered the freezing/melting point of the water, but sugar does not make ice melt as fast as salt does.
Ice cream is a mixture of milk, cream, sugar, and sometimes other ingredients that has been frozen into a When the melting point of ice is lowered, it draws heat out from the ice-cream mixture faster The salt that is mixed with the ice never comes in contact with the ice cream and therefore does not...
When ice melts, it absorbs as much energy as it would take to heat an equivalent mass of water by 80 °C. During the melting process, the temperature remains constant at Other colors can appear in the presence of light absorbing impurities, where the impurity is dictating the color rather than the ice itself.
Why Salt Melts Ice. Usually water freezes at 32° F (0° C), but when it is mixed with salt it lowers the freezing point significantly. Just to clarify, salt lowers the freezing point of water, but it actually doesn't do the melting. The ice melts because of the water around it that is warmer than it is.
Sugar Dissolves Faster Than Salt in Water. This experiment is suitable for early elementary students and will test the solubility of salt and sugar. Why Does Salt Melt Ice Faster Than Sugar?
Table of Contents Does Epsom salt melt ice? Why does salt melt ice faster than sugar? The salt or sugar in an ice cube absorbs the surrounding heat energy faster than frozen water.
A quick google search gave this result for "Why does salt melts ice": "In its pure state, water freezes at 0°C or 32°F. By using salt, that freezing point can be lowered which forces the ice to melt and prevents the water from freezing or...
Why are ice sheets important? Ice sheets contain enormous quantities of frozen water. If the Greenland Ice Sheet melted, scientists estimate that sea level would However, the decline continues to outpace accumulation because warmer temperatures have led to increased melt and faster glacier movement...
With technology moving faster than ever, you’d think there would be an easier way than sticking your finger every few hours and then figuring out how much insulin to inject. There is good news for those with Type 1 diabetes, science and technology have finally come together to create the artificial pancreas.
The ice loss from Greenland and Antarctica is tracking the worst-case climate warming scenario set out by the Intergovernmental Panel on Climate Change The average annual loss of ice from Greenland and Antarctica in the 2010s was 475bn tonnes - six times greater than the 81bn tonnes a year lost in...
Why Salt? Ice forms when the temperature of water reaches 32 degrees Fahrenheit (0 degrees Celsius), and that includes ice on roadways. For one, rock salt does have its limits. If the temperature of the roadway is lower than about 15 degrees F (- 9 C), the salt won't have any effect...
Does ice melt faster in fresh or salt water? Made for parents and teachers My Filming equipment: iPhone 8 ... We want to find out if and why salt water melts ice faster than tap water. Stay tuned fam.
How does salt melt ice? It's a matter of simple chemistry. Salt is an excellent ice-melter, but have you ever asked yourself, "Why does salt melt ice?" Alfalfa meal. Coffee grounds. Sugar beet juice. Heated snow-melting mats. Since salt can't always melt the ice on roads in colder climates...
Why does salt melt ice? In pure water, at 0°C, ice melts just as fast as water freezes. You won't see any of the ice melt as long as the freezing rate What is happening is that the salt is causing the ice to melt but the process of melting requires a lot more energy than raising it's temperature by a degree.













/Salt-sidewalk-58ebea1f5f9b58ef7e8501d8.jpg)






/two-glasses-full-of-crushed-ice-with-frost-on-outside-of-one-melting-ice-below-and-heap-of-salt-98358220-58a4c7953df78c345b909525.jpg)













0 Response to "39 why does salt melt ice faster than sugar"
Post a Comment